Flue Gas Desulfurization Technologies
Flue Gas Desulfurization (FGD) is a process used to remove sulfur dioxide (SO2) from the flue gases generated by various industrial plants, such as power plants, refineries, smelting facilities, and waste incineration facilities. SO2 is an acid gas that can have harmful effects on the environment and human health if released into the atmosphere.
There are different methods to conduct FGD, depending on the gas characteristics and the desired efficiency. These methods include:
- Wet Scrubber: This method uses an alkaline solution as a sorbent, such as lime, caustic soda, ammonia, or seawater, to scrub gases.
- Sulphur, Sulfuric acid/Fertilizer process: This method recovers SO2 in the form of commercial quality materials such as sulphur, sulphuric acid, and fertilizers.
- SNOX Flue gas desulfurization: This method removes not only sulphur dioxide but also nitrogen oxides and particulates from flue gases.
- Scrubbing with an alkali solid or solution.
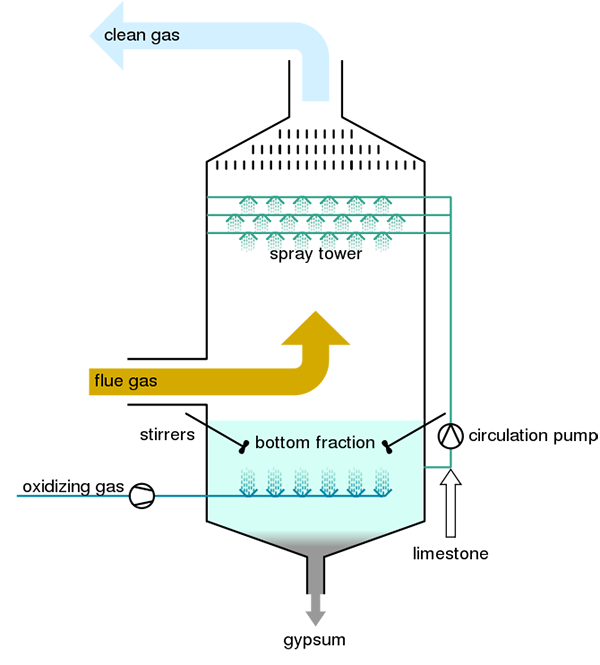
The working principle of FGD is to use an alkaline sorbent slurry or other materials to remove SO2 from flue gases. When wet scrubbing with a CaCO3 (limestone) slurry, for example, the reaction produces calcium sulphite (CaSO3) and carbon dioxide (CO2), which can be expressed in the simplified dry form as:
CaCO3(s) + SO2(g) → CaSO3(s) + CO2(g)
Similarly, when wet scrubbing with a Ca(OH)2 (hydrated lime) slurry, the reaction produces calcium sulphite (CaSO3) and water (H2O), which can be expressed in the simplified dry form as:
Ca(OH)2(s) + SO2(g) → CaSO3(s) + H2O(l)
Other materials, such as magnesium hydroxide (Mg(OH)2), can also be used as sorbents.
To offset the cost of FGD installation, some designs, arid sorbent injection systems, further oxidize the CaSO3 (calcium sulphite) to produce marketable CaSO4-2H2O (gypsum), which can be used in wallboard and other products. The process by which this synthetic gypsum is created is known as forced oxidation:
CaSO3(aq) + 2H2O(l) + ½O2(g) → CaSO4 · 2H2O(s)

Seawater can also be used as a natural alkaline to absorb SO2. The SO2 is absorbed in the water, and when added oxygen reacts to form sulphate ions (SO42-) and free hydrogen ions (H+). The excess H+ is offset by the carbonates in seawater, pushing the carbonate equilibrium to release CO2 gas:
SO2(g) + H2O(l) + ½O2(g) → SO42-(aq) + 2H+(aq)
HCO3-(aq) + H+(aq) → H2O(l) + CO2(g)
In industry, caustic (NaOH) is often used to scrub SO2, producing sodium sulphite:
2NaOH(aq) + SO2(g) → Na2SO3(aq) + H2O(l)
Various wet scrubber designs have been used to achieve maximum gas-liquid surface area and residence time, including spray towers, ventures, and plate towers
Flue Gas Desulfurization (FGD) technologies offer several benefits, including
Emission Reduction
FGD systems help in reducing sulfur dioxide (SO2) emissions from power plants and industrial processes. This helps in improving air quality and mitigating the adverse effects of SO2 on human health and the environment.
Compliance with Regulations
FGD technologies enable companies to meet regulatory standards and emissions limits set by environmental agencies. By implementing FGD systems, businesses can avoid penalties and ensure compliance with emissions regulations.
Acid Rain Prevention
SO2 emissions contribute to the formation of acid rain, which can be harmful to ecosystems, damage buildings, and impact water quality. FGD technologies help in removing sulfur dioxide from flue gases, thereby reducing the formation of acid rain.
Improved Public Health
SO2 is a major contributor to respiratory issues and can aggravate existing health conditions such as asthma. By reducing SO2 emissions through FGD technologies, the overall air quality improves, leading to better public health outcomes.
Protection of Infrastructure
Acid rain resulting from SO2 emissions can cause corrosion of buildings, bridges, and other infrastructure. FGD systems mitigate the impact of acid rain, protecting vital structures and extending their lifespan.
Sustainable Operations
FGD technologies promote sustainable practices by reducing the environmental impact of industrial activities. By implementing FGD systems, companies can demonstrate their commitment to environmental stewardship and sustainability.
These benefits make FGD technologies an important consideration for industries seeking to reduce emissions and comply with environmental regulations.
Technical features of Flue Gas Desulfurization (FGD) technologies include
- Compact equipment design and small footprint
- Strong adaptability to various feed specifications
- High desulfurization efficiency that can reach over 95%
- Use of limestone as an absorbent, which is cheap and easily obtainable
- Ability to use other industrial bases as absorbents
- Mature technology with high reliability
