A lime dosing system must have good storage, the right pump and pipes, and advanced dosing sensors. The water treated with lime has a better odour and is safe for drinking, irrigation, and more.
5 Best Advanced Water Treatment Technologies
Clean water is necessary for drinking, cooking, industry, farming, etc. But sometimes the water has different types of pollutants that make it harmful for humans, plants, and animals. The most common contaminants found in water worldwide are dirt, bacteria, chemicals, salts, microscopic pollutants, and, worst of all, compounds such as pharmaceuticals (hormones and NSAIDs) and industrial chemicals.
Most of the time, traditional water treatment methods (like filtration, settling, or simple disinfection) cannot remove all these contaminants. They are sometimes very small or dissolved in the water and cannot be seen with the naked eye, so we need better treatment methods. In this article, we explain advanced water treatment technologies and how they help us have safer water and a cleaner environment.
What Is Advanced Water Treatment?
Advanced water treatment means using modern facilities and multiple steps to clean water to a much higher standard. The aim of this procedure is to remove large contaminants and microscopic ones, dissolved chemicals, nutrients, toxic materials, etc. Of course, the taste and smell of the water are also improved if it is going to be used for drinking.
In other words, imagine water passes through many filters, and each one removes a different type or size of contaminant. By doing so, the engineers make sure that if one filter misses a pollutant, the next will catch it. Depending on the water treatment plant, these layers might include:
- Physical separation (filtering larger particles, AKA gross contaminants)
- Chemical treatment (adding chemicals to balance the water’s pH, coagulate pollutants, etc.)
- Biological treatment (using helpful bacteria)
- Oxidation (breaking the organic compounds)
- Nanofiltration membranes (only let water pass)
5 Advanced Water Treatment Technologies
Here are 5 advanced water treatment technologies used in many developed countries.
a) Membrane filtration
Membrane filters are like sieves that remove pollutants from the water. Depending on the size of the particles or pollutants, one of the following 4 types of membrane filtration is used.
- Reverse osmosis (0.1 nanometer)
Reverse osmosis is a semi-permeable membrane that removes salts and other small contaminants from the water. In this technique, the water enters the filter with high pressure, so the salt, bacteria, and other pollutants are kept behind the membrane. The RO then creates two streams, one with clean water that can be used for drinking and irrigating the plants, and one with polluted water that should be refined again.

- Nanofiltration (1 to 10 nanometers)
Nanofiltration membranes are similar to RO, but their pores are a bit larger. It removes small organic molecules, pesticides, some salts, and dissolved metals and ions (like calcium and magnesium). In this technique, too, the water enters the system with pressure, and its impurities are trapped behind the filter. Unfortunately, the filter allows very small molecules, such as sodium and chloride, to pass through; however, it is best for removing industrial acids.
- Ultrafiltration (0.01 to 0.1 microns)
The pores in ultrafiltration are larger than in naofilters. This type of filter is used to remove small suspended solids and heavy molecules. The wastewater is pressurised and pumped into the system; otherwise, it would not pass the filter’s microscopic pores. This method requires less energy and chemicals than the others, while its output is excellent.
4. Microfiltration (0.1 to 10 microns)
This type of filter is usually used to catch large particles from the water, such as algae, dirt, and debris.
b) Adsorption and media filters
The second advanced water treatment method is adsorption. In this method, granular activated carbon (GAC), activated alumina, or other materials (like silica gel, zeolites, etc.) are used to remove pesticides, viruses, asbestos, organic compounds, and chlorine from the wastewater. GAC is made of heated coconut shells or coal and has lots of large and small pores that trap organic chemicals, taste/odour compounds, and others.
Although the adsorption and media filters improve the water’s taste, odour, and clarity, they don’t reduce nitrates, fluoride, or microorganisms in the water. Also, the activated carbon gets saturated fast and needs to be replaced.

C) Advanced oxidation processes
Some contaminants are stubborn, so it’s hard to remove or break them down. That’s where advanced oxidation processes (AOPs) are needed. AOPs are chemical and air treatment techniques that use reactive radicals (·OH) to turn organic or some inorganic pollutants into harmless products (i.e., water, carbon dioxide).
These advanced water treatment techniques are usually used when the water contains pharmaceutical residues, microplastics, endocrine disruptors, pesticides, etc. The engineers then use oxidants like ozone or hydrogen peroxide as well as UV radiation, ultrasound, or electricity to refine the water. Common AOP methods are:
- Ozone-based processes
- Photocatalysis
- Fenton and photo-Fenton processes
- Electrochemical oxidation
- Sonolysis
- A combination of AOPs with biological treatment methods (the most cost-effective)
d) Biological treatment
In wastewater treatment, biological treatment is the most cost-effective and eco-friendly method because the bacteria naturally consume organic substances or nutrients (nitrogen, phosphorus). In advanced water treatment, the engineers use biofilters or biological reactors that remove nutrients or leftover organic compounds. This helps reduce nutrients that could help algae grow faster. Note that advanced biological treatment is usually combined with other physical or chemical processes (like activated carbon or advanced oxidation processes) for better results.
e) Other advanced technologies
Besides the above-mentioned systems, there are some less-used water treatment technologies such as flow-electrode capacitive deionisation (FCDI), shock electrodialysis, and photodegradation. These eco-friendly technologies:
- Remove dissolved ions (salts)
- Desalinate water
- Filter particles
- Disinfect water
- Remove micropollutants
7 Steps of Advanced Water Treatment
Usually, the wastewater goes through the following 7 stages to have a standard quality again.
|
Stage |
Purpose |
|
Pretreatment |
Remove large debris (such as leaves, trash, cigar butts, etc.) so other equipment isn’t damaged. |
|
Physical filtration |
Remove suspended solids or smaller particles through microfiltration and ultrafiltration. |
|
Membrane filtration/desalination |
Remove dissolved salts or micro contaminants through nanofiltration or reverse osmosis (RO). |
|
Chemical/oxidation polishing |
Use advanced oxidation or chemicals to break down stubborn organic chemicals or micropollutants. |
|
Biological polishing |
Remove nutrients or remaining organics via microbial processes. |
|
Final disinfection |
Use UV, ozone, etc. to kill any remaining microorganisms or pathogens. |
| Post-treatment |
Adjust water quality (pH, hardness, taste, odour) and remove residual chemicals before reusing it. |
Benefits of Advanced Water Treatment
Advanced water treatment technologies help us in many ways. Here are some examples.
- Safer drinking water: advanced treatment helps remove chemicals, micropollutants, pathogens, etc. from the water, so it makes the water safe for drinking.
- Water reuse: treated water can be reused for irrigation, cooling industrial equipment, generating steam, or even for drinking.
- Meeting stricter regulations: day by day, the laws get stricter about water quality. Advanced methods help keep up with these standards.
- Protecting the environment: by removing nutrients (nitrogen, phosphorus) or harmful chemicals, treated water is safer to release back into rivers, lakes, or groundwater.
Challenges and Drawbacks of Advanced Water Treatment
Nothing is perfect, not even advanced water treatment technologies that keep our universe safe and livable. They have 3 challenges:
- Installing, maintaining, and replacing membranes, oxidation systems, and other equipment is costly. Also, these systems require skilled engineers and operators, whose salaries are quite high.
- High pressures and electrical or UV systems consume too much energy.
membranes can get clogged, filters need cleaning or replacement, oxidation chemicals need control, and activated carbon needs regeneration. Maintaining all these systems is time-consuming. - Some methods leave waste brine, concentrated contaminants, or chemical byproducts behind. Disposing of these sludges is hard and costly.
Also Read: Excellent Techniques for Reducing Sludge in Wastewater Plants

A Quick Review
Advanced water treatment is one step further than basic cleaning. It is a combination of seven steps that remove small, dissolved, or stubborn pollutants from the water and make it reusable. The most common water treatment technologies are membrane filtration, adsorption, chemical oxidation, and biological treatment. Although advanced water treatment is costlier and more complex, its results are marvellous. At AquipTech (AIMEQUIP), we make sure the water meets the latest standards and is healthy enough to be used for drinking and irrigation.
FAQs
- Can water treatment technologies be used for seawater?
In remote or drought-prone regions, the engineers use desalination (RO) or advanced membranes + oxidation to make seawater drinkable.
- Which membrane filtration is the best?
There is no best membrane filtration. They all serve different purposes; for example, reverse osmosis is good for removing salt and bacteria, nanofiltration is best for softening water, ultrafiltration removes larger particles like microorganisms, and microfiltration is used for removing very large pollutants.
- Can solar energy be used to treat water?
Yes. Solar Water Disinfection (SODIS) is a type of advanced water treatment that uses the sun’s UV radiation and heat to kill pathogens. This method is very eco-friendly and cheap compared to other methods.
Chemical Dosing System: The Process + 8 Best Chemicals
The chemical dosing system adjusts the water pH, disinfects the wastewater, prevents algae formation, and more. Depending on the size and purpose of chemical dosing, different types of pumps are used, such as piston, Peristaltic, and diaphragm ones.
Chemical Treatment of Wastewater: 7 Critical Steps
Chemical wastewater treatment has 7 steps, from equalizing the incoming water and adjusting its pH to disposing of its sludge. All these steps are necessary to make the wastewater clean and safe again, especially for reuse.
Runoff Pollution: 5 Shocking Causes + Best Solutions
The main causes of runoff pollution are impervious surfaces, land‐use change, uncontrolled soil erosion, and low-absorptive soil. However, the stormwater runoff can be prevented by using less chemical fertilizers and pesticides, cleaning storm drains regularly, planting native plants, etc.
Drilling Mud Disposal: 5 Brilliant Techniques Everyone Must Know
In large projects, like creating wells for oil and gas extraction, drilling mud is pumped into boreholes to cool down the drill bit, grease the drill, carry rock cuttings to the surface, support the wellbore, and control the pressure. Drilling mud can contaminate the soil and water if left untreated, especially when they are mixed with toxins and chemicals. Therefore, they should be cleaned of harmful substances before returning to the environment. In the following text, we discuss different techniques of drilling mud disposal, explain how drilling mud puts the environment at risk, and more.
What is Drilling Mud?
Drilling mud is a thick liquid used in drilling operations. Its base ingredient is water (most common), oil, or gas; however, the engineers must add bentonite clay and barite to balance its density and viscosity. There are different types of drilling muds depending on the materials used and the purpose of the drilling. Unfortunately, when drilling ends, this mud becomes waste, and disposing of it is a big challenge.
3 Types of Drilling Muds (Based on Their Base Ingredient)
1. Oil-Based
The main ingredient in oil-based muds is mineral or synthetic oils. Oil is usually used in drilling very deep wells, as it lubricates the drill bits better. But these muds are usually toxic, flammable, and non-biodegradable.
The best disposal methods:
- Thermal treatment or incineration to destroy hydrocarbons.
- Removing solids and recovering the base oil for further use.
2. Water-Based
In water-based muds, fresh water, seawater, or brine is the main component. The engineers usually add bentonite clay and barite to increase its density, and other chemicals to control its viscosity. Although water-based muds are less harmful than oil-based ones, they still contain chemicals and heavy metals.
The best disposal methods:
- Landfarming and bioremediation to break down pollutants.
- Dewatering and pit burial.
3. Air-Based
Air-based drilling mud, also known as aerated or pneumatic drilling fluid, uses compressed gas instead of water or oil. Pneumatic drilling fluid is cheaper and super eco-friendly, but it is only suitable for extremely hard and dry rocks.
The best disposal methods:
- Pit burial.
- Reuse in construction or land reclamation if the toxins are removed.

The Best 5 Techniques for Drilling Mud Disposal
Drilling mud removal must be done carefully to avoid soil, air, and water contamination. The best disposal techniques in 2025 include:
1. Landfarming
This is the most cost-effective and energy-efficient method of drilling mud disposal. It is a simple procedure, but it only works for non-hazardous, water-based muds. Landfarming involves two main steps:
a) The waste mud is spread on a large, well-prepared land. Then, the machines mix it with topsoil.
b) The microbes break down the organic materials into less harmful materials, such as carbon dioxide, water, and biomass.
2. Pit Burial or Landfill
In this method, the mud is first dried through natural evaporation, chemical solidification, or aeration. When the waste is completely dewatered, it is buried in the same pit or in an adjacent well. Finally, the burial pit is covered with soil to return the land to its natural state. The cover also prevents animals from scavenging and blocks disease spread.
3. Thermal Desorption
This is an expensive method of removing drilling mud, yet it is the best one to get rid of oils and chemicals. After separating large stones, the remaining mud is heated in a kiln to 500°C. This procedure recovers hydrocarbons from the mud. The vaporised hydrocarbon is then reused in later drilling operations.
4. Recycling and Reuse
Some muds, especially oil-based ones, can be cleaned and reused after removing large particles and contaminants. Shakers, hydrocyclones, and centrifuges are usually used to refine and return the fluid to the drilling operations.
5. Solidification/Stabilisation (S/S)
In this technique, binding agents like cement, fly ash, or lime are used to turn liquid mud into a solid waste. The dry material can then be used in construction or be buried in the pit burials.

How Does Untreated Drilling Mud Impact the Environment?
Improper drilling mud disposal can damage the ecosystem and wildlife. Oil-based muds affect the plant’s growth, harm aquatic life, and reduce soil fertility. Some wastes also contain toxic metals like barium, chromium, or lead, which can remain in the soil for decades. In addition to the environment, untreated drilling mud might put workers’ lives at risk. Skin irritation, respiratory issues, headaches, and nausea are the most reported health issues by the exposed workers.
What Type of Drilling Mud Is Hazardous?
Drilling mud is classified as hazardous if it:
- Contains high levels of toxic substances (like oil, heavy metals, or sulphides).
- Is flammable, corrosive, or reactive.
- Fails environmental safety tests, such as the Toxicity Characteristic Leaching Procedure (TCLP).
In general, oil-based mud (both synthetic and mineral) is the most hazardous type of waste mud because it is extremely flammable and contains high levels of toxins, such as benzene and xylene. On the other hand, water-based mud is the least hazardous one, unless it is mixed with contaminants from the wellbore, especially hydrogen sulphide.
The Last Words
Drilling mud keeps drilling operations safe and effective, but what happens to its waste is also important. The way we handle drilling mud removal can make a huge difference for our environment. If oil and chemicals leak into the soil or water sources, the nearby flora and fauna will be damaged. So, we must make sure today’s work doesn’t cause tomorrow’s problems. By using smarter methods like recycling and treatment, we can reduce mud waste and keep our surroundings clean. AquipTech (AIMEQUIP) is one of the top Australian companies focusing on wastewater treatment. With its advanced engineering solutions and industrial machinery, it helps protect the planet we live on.
Also Read: Advanced Water Treatment Technologies: Enhancing Efficiency and Sustainability
FAQs about Drilling Mud Disposal
1. Why is drilling mud disposal important?
Wrong drilling mud disposal can pollute soil, water, and air, and increase the health and environmental hazards. It can also negatively affect the flora and fauna of the area.
2. Can drilling mud be recycled?
Yes. Many components can be recovered and reused. Of course, recycling is mostly used for oil-based and air-based muds.
3. Which type of drilling mud is most harmful to the environment?
Oil-based muds are the most harmful because they contain hydrocarbons that are toxic, extremely flammable, and non-biodegradable.
4. What happens if drilling mud leaks into the ground?
It can contaminate groundwater and lower soil fertility. To prevent such problems, the engineers place a membrane at the bottom of the burial pits. In some cases, they also divide the buried waste into two parts and place a geo-membrane between them. In this way, they can detect any leakage that might occur.
Excellent Techniques for Reducing Sludge in Wastewater Plants
Reducing sludge in wastewater plants involves 5 techniques: 1) using anaerobic or hybrid systems, 2) settling the sludge by controlling filamentous bacteria, and 3 other methods.
6 Dangerous Challenges of Wastewater Treatment Basins in Australia
Wastewater Treatment Basins in Australia face some serious challenges that can’t be overlooked. Their parts age and corrode, sediments build up at their bottom, and discharging their water requires time and advanced tools, etc.
7 Critical Wastewater Treatment Challenges in Australia You Can’t Ignore
The most common wastewater treatment challenges in Australia are improper initial filtering, a lack of nitrogen treatment, failure to lower BOD, and more. The basins also face serious challenges in Australia, like ageing, wearing out, corroding silently, getting too hot due to heat waves, etc.
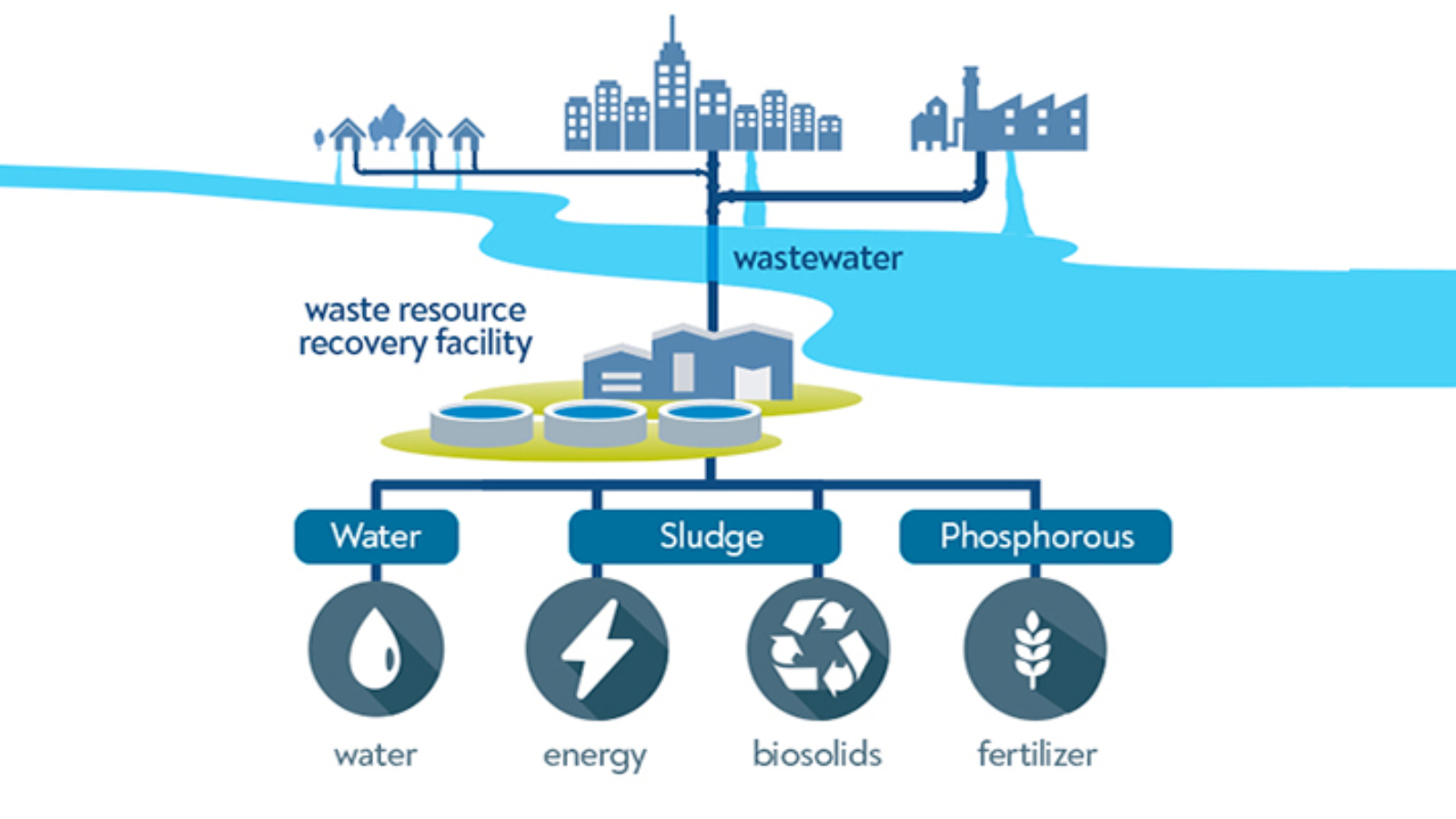
Turning Waste into Resources: Innovative Wastewater Recovery Solutions
Water is one of the most valuable resources on our planet, yet industries and communities continue to generate vast amounts of wastewater every day. Traditionally, wastewater was seen only as a problem to be treated and discharged. Today, with the growing need for sustainability, wastewater recovery has emerged as an innovative approach that transforms waste into valuable resources.
At AIMEQUIP, we specialise in advanced wastewater treatment and recovery solutions that not only protect the environment but also unlock economic and operational benefits for industries across Australia.
What is Wastewater Recovery?
Wastewater recovery is the process of treating wastewater in a way that allows valuable resources, such as clean water, nutrients, and even energy to be recovered and reused. Unlike conventional wastewater treatment, which focuses mainly on safe disposal, wastewater recovery prioritises reuse, recycling, and the circular economy.
Industries such as mining, food processing, manufacturing, and municipal utilities are increasingly turning to wastewater recovery solutions to reduce costs, minimise environmental impact, and achieve compliance with strict regulations.
Benefits of Wastewater Recovery
Environmental Benefits
- Reduces the volume of pollutants discharged into rivers, lakes, and oceans.
- Supports sustainable water management by lowering demand for freshwater extraction.
- Contributes to global and local climate goals by reducing carbon emissions.
Economic Benefits
- Cuts operational costs by reducing the need to purchase fresh water.
- Minimises wastewater disposal fees and penalties.
- Opens new opportunities for revenue generation through recovered energy and nutrients.
Social & Regulatory Benefits
- Helps industries meet government regulations and environmental standards.
- Enhances corporate sustainability initiatives, improving brand reputation.
- Protects community water resources for future generations.
Innovative Wastewater Recovery Technologies
Modern wastewater recovery solutions use advanced technologies to maximise efficiency and sustainability:
- Membrane Filtration & Advanced Oxidation: Produces high-quality water for reuse.
- Nanobubble and Aeration Systems: Improves oxygen transfer, enhances treatment, and supports sludge breakdown.
- Chemical Dosing and Flocculant Dosing Systems: Essential for effective coagulation, sedimentation, and contaminant removal. (AIMEQUIP specialises in these solutions to optimise recovery performance.)
- Energy and Nutrient Recovery Systems: Converts sludge into biogas and extracts valuable nutrients like phosphorus and nitrogen for fertiliser production.
Case Studies: Wastewater Recovery in Action
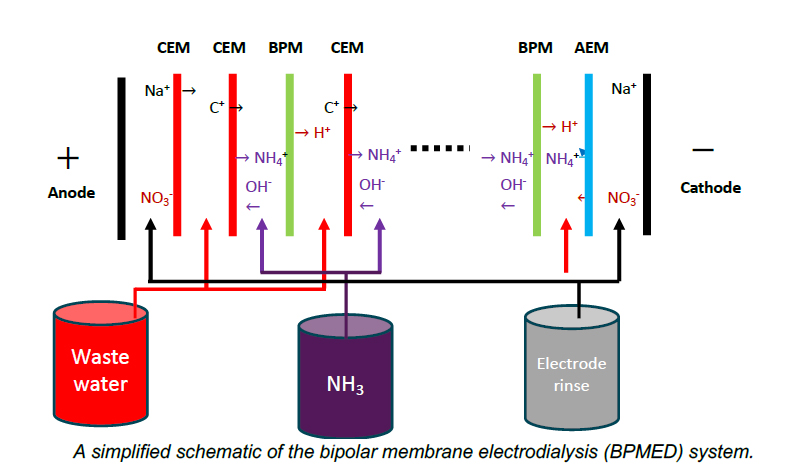
1- Ammonia Recovery with Bipolar Membrane Electrodialysis (CSIRO, WA)
A study by CSIRO explored ammonia recovery from wastewater using bipolar membrane electrodialysis (BPMED). The process achieved up to 90% ammonia recovery from anaerobic sludge centrate, transforming a waste pollutant into a valuable energy carrier. Ammonia can be reused as a fuel, for hydrogen production, or as a green chemical feedstock. This approach highlights how wastewater recovery reduces treatment costs, supports greenhouse gas reduction, and enables a circular economy initiative.
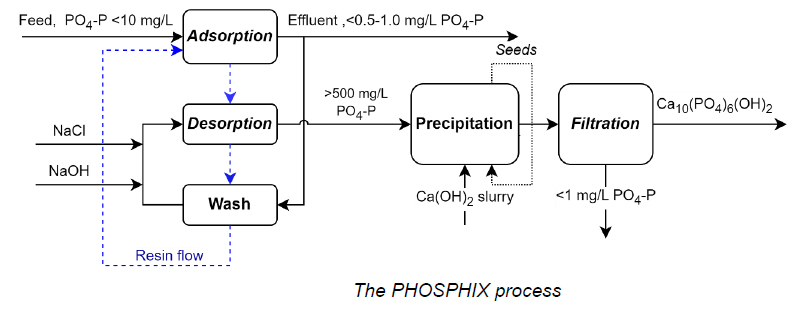
2- Phosphorus Recovery with Continuous Ion Exchange (Clean TeQ Water, VIC)
Clean TeQ Water developed the PHOSPHIX® process, which uses continuous ion exchange to remove and recover phosphate from wastewater. In pilot studies, phosphate levels were reduced to below 1 mg/L, while phosphorus was recovered as hydroxyapatite, a material that can be reused as fertiliser. The process not only helps industries comply with strict discharge limits but also creates a new revenue stream by producing a marketable byproduct. This is a practical example of how nutrient recovery can transform compliance costs into economic and environmental value.
3- Copper Removal from Water Treatment Residuals (SUEZ & SA Water, SA)
In South Australia, SUEZ and SA Water trialled a project to recover copper from water treatment sludge. Using an adapted acid leaching process, they successfully reduced waste classification levels while producing a copper-rich solution with reuse potential. The trial demonstrated that high-level contaminated waste could be reduced by up to 48% in disposal cost. By diverting valuable copper away from landfill and back into circulation, this project shows how waste stream collaboration supports both cost savings and resource recovery.
Why These Case Studies Matter
These projects prove that wastewater recovery is more than theory—it’s happening now across Australia. From ammonia and phosphorus recovery to heavy metal reuse, each case highlights how turning waste into resources:
- Reduces operational costs
- Supports regulatory compliance
- Creates new revenue opportunities
- Contributes to Australia’s circular economy goals
At AIMEQUIP, we bring this same mindset into every wastewater recovery project, delivering tailored solutions that transform waste into valuable assets.
The Future of Wastewater Recovery
The future of water management is rooted in the circular economy, where waste becomes a resource instead of a burden. Globally, wastewater recovery is expected to grow rapidly as industries and governments recognise its importance in securing water supplies and achieving carbon reduction targets.
In Australia, companies like AIMEQUIP are at the forefront of this transformation, helping industries turn challenges into opportunities through innovative wastewater recovery solutions.
At AIMEQUIP, we are proud to provide advanced wastewater treatment and recovery solutions that help industries improve efficiency, reduce costs, and contribute to a more sustainable future. Contact AIMEQUIP today to learn how our wastewater recovery systems can support your business in turning waste into resources.